Matter is everything around you. Atoms and molecules are all composed of matter. Matter is anything that has mass and takes up space. If you are new to the idea of mass, it is the amount of stuff in an object. We talk about the difference between mass and weight in another section. Matter is sometimes related to light and electromagnetic radiation.
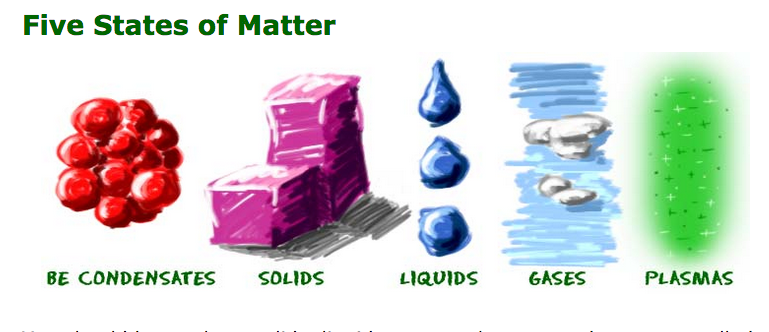
Even though matter can be found all over the Universe, you will only find it in a few forms on Earth. We cover five states of matter on the site. Each of those states is sometimes called a phase. There are many other states of matter that exist in extreme environments. Scientists will probably discover more states as we continue to explore the Universe.
Even though matter can be found all over the Universe, you will only find it in a few forms on Earth. We cover five states of matter on the site. Each of those states is sometimes called a phase. There are many other states of matter that exist in extreme environments. Scientists will probably discover more states as we continue to explore the Universe.
You should know about solids, liquids, gases, plasmas, and one state called the Bose-Einstein condensate (BEC). Scientists have always known about solids, liquids, and gases. Plasma was a new idea when it was identified by William Crookes in 1879. The scientists who worked with the Bose-Einstein condensate received a Nobel Prize for their work in 1995.
What makes a state of matter? It's about the physical state of the molecules and atoms. Think about solids. They are often hard and brittle. Liquids are fluidy, can move around a little, and fill up containers. Gases are always around you, but the molecules of a gas are much farther apart than the molecules in a liquid. If a gas has an odor, you’ll be able to smell it before you can see it. The BEC is all about atoms that are even closer and less energetic than atoms in a solid.
What makes a state of matter? It's about the physical state of the molecules and atoms. Think about solids. They are often hard and brittle. Liquids are fluidy, can move around a little, and fill up containers. Gases are always around you, but the molecules of a gas are much farther apart than the molecules in a liquid. If a gas has an odor, you’ll be able to smell it before you can see it. The BEC is all about atoms that are even closer and less energetic than atoms in a solid.
Molecules can move from one physical state to another and not change their basic structure. Oxygen (O2) as a gas has the same chemical properties as liquid oxygen. The liquid state is colder and denser, but the molecules (the basic parts) are still the same. Water (H2O) is another example. A water molecule is made up of two hydrogen (H) atoms and one oxygen (O) atom. It has the same molecular structure whether it is a gas,liquid, or solid. Although its physical state may change, its chemical state remains the same.
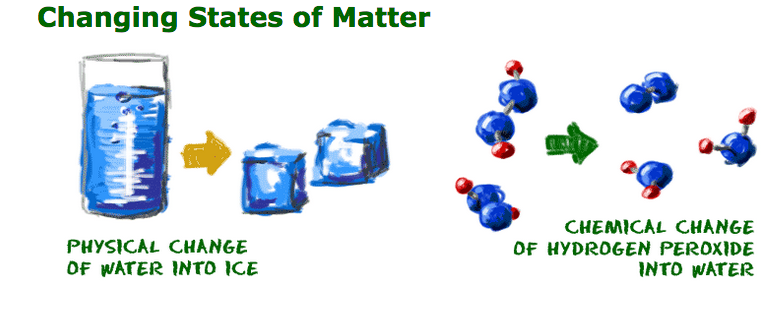
So you're asking, "What is a chemical change?" Let's start with a glass of pure water. If the formula of water were to change, that would be a chemical change. If you could add a second oxygen atom to a water molecule, you would have hydrogen peroxide (H2O2). The molecules would not be water anymore. The reality of creating hydrogen peroxide is more difficult.
Chemical changes occur when the bonds between atoms in a molecule are created or destroyed. Changes in the physical state are related to changes in the environment such as temperature, pressure, and other physical forces. Generally, the basic chemical structure does not change when there is a physical change. Of course, in extreme environments such as the Sun, no molecule is safe from destruction.
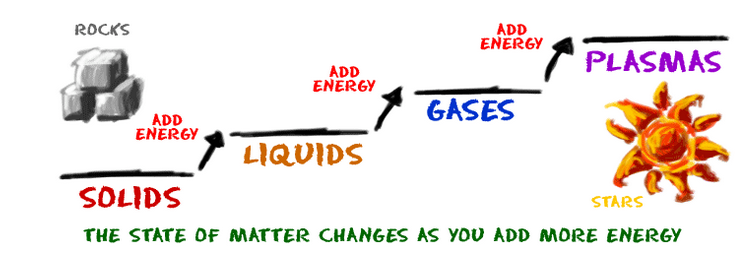
We look at five states of matter on the site. Solids, liquids, gases, plasmas, and Bose-Einstein condensates (BEC) are different states that have different physical properties. Each of these states is also known as a phase. Elements and compounds can move from one phase to another when specific physical conditions change. For example, when the temperature of a system goes up, the matter in the system becomes more excited and active. If enough energy is placed in a system, a phase change may occur as the matter moves to a more active state.
Think about it this way. Let’s say you have a glass of water (H2O). When the temperature of the water goes up, the molecules get more excited and bounce around a lot more. If you give a liquid water molecule enough energy, it escapes the liquid phase and becomes a gas.
Have you ever noticed that you can smell a turkey dinner after it starts to heat up? As the energy of the molecules inside the turkey heat up, they escape as a gas. You are able to smell those volatile molecules that are mixed in the air.
It’s About the Physical "Phase" describes a physical state of matter. The key word to notice is physical. Things only move from one phase to another by physical means. If energy is added (like increasing the temperature) or if energy is taken away (like freezing something), you have created a physical change.
So you're asking, "What is a chemical change?" Let's start with a glass of pure water. If the formula of water were to change, that would be a chemical change. If you could add a second oxygen atom to a water molecule, you would have hydrogen peroxide (H2O2). The molecules would not be water anymore. The reality of creating hydrogen peroxide is more difficult.
Chemical changes occur when the bonds between atoms in a molecule are created or destroyed. Changes in the physical state are related to changes in the environment such as temperature, pressure, and other physical forces. Generally, the basic chemical structure does not change when there is a physical change. Of course, in extreme environments such as the Sun, no molecule is safe from destruction.
We look at five states of matter on the site. Solids, liquids, gases, plasmas, and Bose-Einstein condensates (BEC) are different states that have different physical properties. Each of these states is also known as a phase. Elements and compounds can move from one phase to another when specific physical conditions change. For example, when the temperature of a system goes up, the matter in the system becomes more excited and active. If enough energy is placed in a system, a phase change may occur as the matter moves to a more active state.
Think about it this way. Let’s say you have a glass of water (H2O). When the temperature of the water goes up, the molecules get more excited and bounce around a lot more. If you give a liquid water molecule enough energy, it escapes the liquid phase and becomes a gas.
Have you ever noticed that you can smell a turkey dinner after it starts to heat up? As the energy of the molecules inside the turkey heat up, they escape as a gas. You are able to smell those volatile molecules that are mixed in the air.
It’s About the Physical "Phase" describes a physical state of matter. The key word to notice is physical. Things only move from one phase to another by physical means. If energy is added (like increasing the temperature) or if energy is taken away (like freezing something), you have created a physical change.
When molecules move from one phase to another they are still the same substance. There is water vapor above a pot of boiling water. That vapor (or gas) can condense and become a drop of water in the cooler air. If you put that liquid drop in the freezer, it would become a solid piece of ice. No matter what physical state it was in, it was always water. It always had the same chemical properties.
On the other hand, a chemical change would build or break the chemical bonds in the water molecules. If you added a carbon (C) atom, you would have formaldehyde (H2CO). If you added an oxygen (O) atom, you would create hydrogen peroxide (H2O2). Neither new compound is anything like the original water molecule. Generally, changes in the physical state do not lead to any chemical change in molecules.
On the other hand, a chemical change would build or break the chemical bonds in the water molecules. If you added a carbon (C) atom, you would have formaldehyde (H2CO). If you added an oxygen (O) atom, you would create hydrogen peroxide (H2O2). Neither new compound is anything like the original water molecule. Generally, changes in the physical state do not lead to any chemical change in molecules.
Changing States of Matter
Solids to Gas and back to Solid
|
You are used to solids melting and becoming liquids. Some of you may have also seen a solid become a gas. It's a process called sublimation. The easiest example of sublimation might be dry ice. Dry ice is solid carbon dioxide (CO2). Amazingly, when you leave dry ice out in a room, it just turns into a gas. Have you ever heard of liquid carbon dioxide? It can be made, but not in normal situations. Coal is another example of a compound that will not melt at normal atmospheric pressures. It will sublimate at very high temperatures.
Can you go from a gas to a solid? Sure. Deposition occurs when a gas becomes a solid without going through the liquid state of matter. Those of you who live near the equator may not have seen it, but closer to the poles we see frost on winter mornings. Those little frost crystals on plants build up when water vapor from the air becomes a solid on the leaves of plants. |
Liquid to Gas and back to Liquid
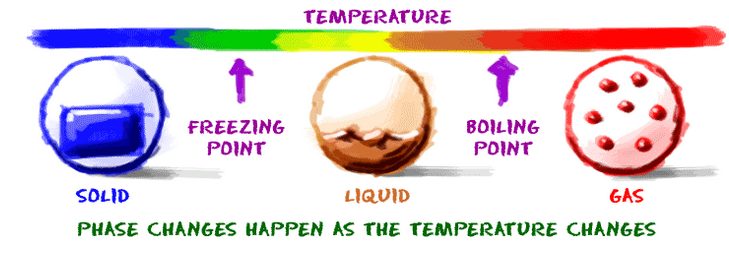
When you are a liquid and want to become a gas, you need to find a lot ofenergy. Once you can direct that energy into your molecules, they will start to vibrate. If they vibrate enough, they can escape the limitations of the liquid environment and become a gas. When you reach your boiling point, the molecules in your system have enough energy to become a gas.
The reverse is true if you are a gas. You need to lose some energy from your very excited gas atoms. The easy answer is to lower the surrounding temperature. When the temperature drops, energy will be transferred out of your gas atoms into the colder environment. When you reach the temperature of the condensation point, you become a liquid. If you were steam over a boiling pot of water and you hit a wall, the wall would be so cool that you would quickly become a liquid. The wall absorbed some of your extra energy.
The reverse is true if you are a gas. You need to lose some energy from your very excited gas atoms. The easy answer is to lower the surrounding temperature. When the temperature drops, energy will be transferred out of your gas atoms into the colder environment. When you reach the temperature of the condensation point, you become a liquid. If you were steam over a boiling pot of water and you hit a wall, the wall would be so cool that you would quickly become a liquid. The wall absorbed some of your extra energy.
Gas to Plasma and back to Gas
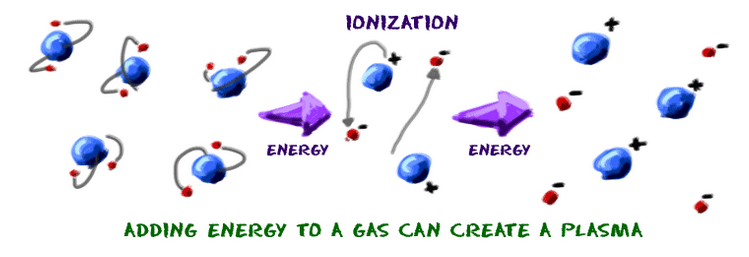
Let's finish up by imagining you're a gas like neon (Ne). You say, "Hmmmm. I'd like to become a plasma. They are too cool!" As a gas, you're already halfway there, but you still need to tear off a bunch ofelectrons from your atoms. The gas needs to ionize. Electrons have a negative charge. Eventually, you'll have groups of positively and negatively charged particles in almost equal concentrations. They wind up in a big plasma ball. Because the positive and negative charges are in equal amounts, the charge of the entire plasma is close to neutral. Neutral happens when a whole bunch of positive particles cancel out the charges of an equal bunch of negatively charged particles.
Plasma can be made from a gas if a lot of energy is pushed into the gas. In the case of neon, it is electrical energy that pulls the electrons off. When it is time to become a gas again, just flip the neon light switch off. Without the electricity to energize the atoms, the neon plasma returns to its gaseous state. We have a special world here on Earth. We have an environment where you don't find a lot of everyday plasma. Once you leave the planet and travel through the Universe, you will find plasma everywhere. It's in stars and all of the space in between.
Plasma can be made from a gas if a lot of energy is pushed into the gas. In the case of neon, it is electrical energy that pulls the electrons off. When it is time to become a gas again, just flip the neon light switch off. Without the electricity to energize the atoms, the neon plasma returns to its gaseous state. We have a special world here on Earth. We have an environment where you don't find a lot of everyday plasma. Once you leave the planet and travel through the Universe, you will find plasma everywhere. It's in stars and all of the space in between.